News
Stay up to date on the latest crypto trends with our expert, in-depth coverage.


Bitcoin drops to $85,000, Dogecoin founder shares unexpected reaction
币界网·2025/12/16 10:14


SLX will launch on Legion on December 22, the Winter Solstice.
BlockBeats·2025/12/16 10:07

CMTA Adopts Chainlink Interoperability Standard for Cross-Chain Tokenized Assets
BlockchainReporter·2025/12/16 10:01

Unshakable Conviction: Why LD Capital’s Founder Sees Strong ETH Fundamentals Amid Market Volatility
Bitcoinworld·2025/12/16 09:57

Bank of America predicts that banks’ transition to blockchain will take years
币界网·2025/12/16 09:52
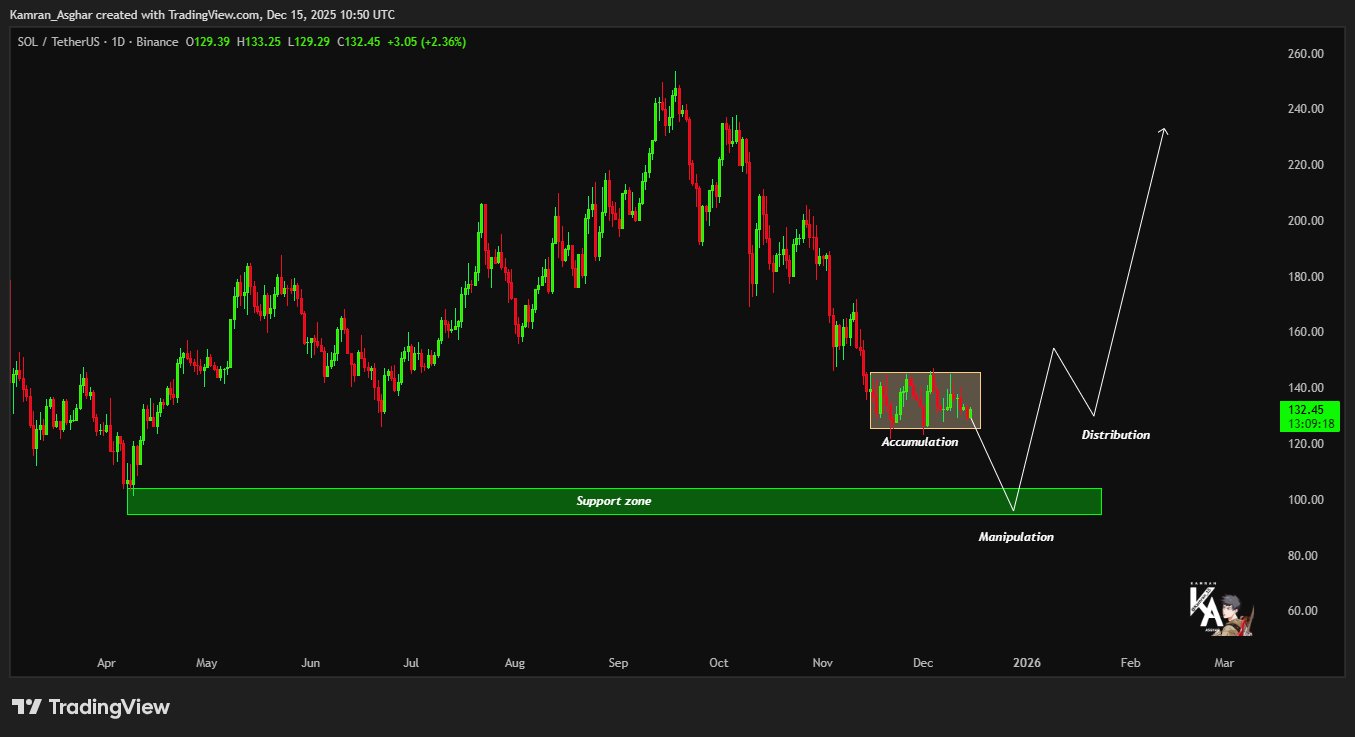
Solana December 16 Price Prediction: Key Level for SOL Price Recovery
币界网·2025/12/16 09:51

MetaMask adds native Bitcoin support, accelerating its multi-chain strategy
币界网·2025/12/16 09:51
Flash
09:25
Yellen: Rebound Fundamentally Over, Now Is the "Darkest Before Dawn"BlockBeats News, May 16th, Liquid Capital (formerly LD Capital) founder Li Hua issued a statement, saying, "This round of rebound is basically over. I have repeatedly emphasized that this round is about observing the rebound instead of a trend reversal, and the strategy is to close long positions on the way up."
A few possible bearish factors in the future include: a normal pullback in the U.S. stock market at its high, the impact of oil prices on inflation leading to changes in interest rate cut expectations, with a focus on a noticeable rebound in U.S. Treasury yields. The long-term outlook for crypto is promising. The current situation is the darkest before the dawn. Every bear market cycle sees numerous pessimists and exits, but deep bear markets often provide good opportunities for entry and patience.
"I suggest allocating 50% of future profits to the AI field, as this is an irreversible trend."
09:23
Yilihua: It is recommended to allocate 50% of future returns to the AI sectorLiquid Capital founder Yilihua posted on X, stating that the current market rebound has basically ended and recommends an upward liquidation strategy. He mentioned possible negative factors including a correction in US stocks at high levels, the impact of oil prices on inflation, and a rebound in US Treasury yields. He believes the crypto market has long-term potential and that this is the darkness before dawn, emphasizing that a deep bear market offers a good opportunity to enter and exercise patience. In addition, he suggests allocating 50% of future returns to the AI sector, considering AI to be an irreversible trend.
09:22
If Bitcoin falls below $76,000, the mainstream CEX long liquidation volume will reach $628 million.BlockBeats News, May 16th, according to Coinglass data, if Bitcoin falls below $76,000, the total long liquidation strength of mainstream CEXs will reach $628 million.
Conversely, if Bitcoin surpasses $80,000, the total short liquidation strength of mainstream CEXs will reach $914 million.
BlockBeats Note: The liquidation chart does not display the exact number of contracts to be liquidated or the precise value of the contracts being liquidated. The bars on the liquidation chart actually represent the importance of each liquidation cluster relative to adjacent liquidation clusters, that is, the strength.
Therefore, the liquidation chart shows to what extent the price of the underlying asset will be affected when it reaches a certain level. A higher "liquidation bar" indicates that the price reaching that level will trigger a more intense reaction due to a liquidity cascade.
News